Practice over 1000+ GATE-level questions from this topic!
Designed to match the latest GATE pattern with topic-wise precision, difficulty tagging, and detailed solutions.
Which of the following can be used as an n-type dopant for silicon?
Select the correct option(s).
Arsenic
Boron
Gallium
Phosphorous
The correct options are:
(A) Arsenic and (D) Phosphorous
These are both 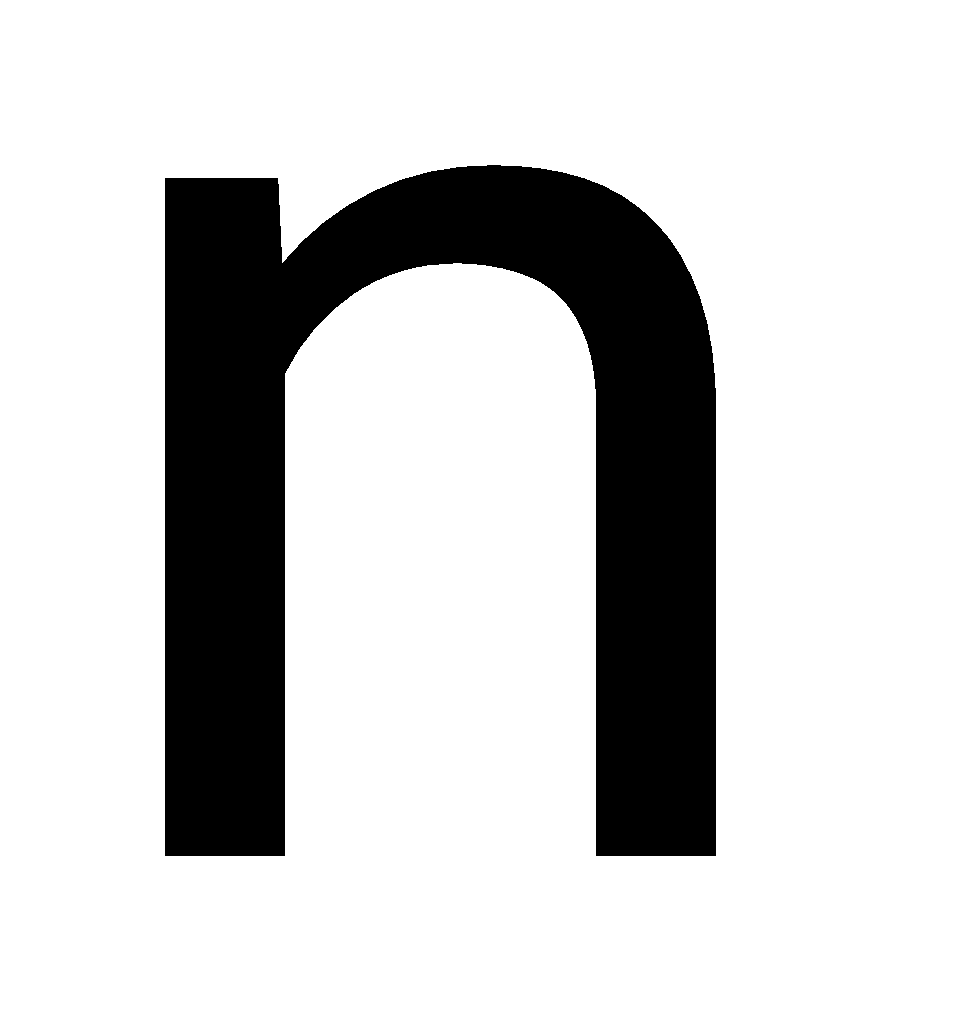 -type dopants for silicon because they have five valence electrons (group
-type dopants for silicon because they have five valence electrons (group  elements), which provide extra electrons to the silicon lattice, making it an n-type semiconductor.
elements), which provide extra electrons to the silicon lattice, making it an n-type semiconductor.
while, Boron and Gallium are p-type dopants because they have only three valence electrons, creating holes in the silicon lattice.
Alternate Solution:
Arsenic (As) and Phosphorous (P) are Group 15 elements, they have an extra electrons (5 electrons in valence shell) to contribute to lattice when doped in silicon.
 n-type means material has more electrons than holes,
n-type means material has more electrons than holes,
n-type dopants are (a) and (d)